What Type Of Defense Includes Animals That Are Colored To Match The Features Of Their Habitat
Abstract
Camouflage is a key defence across taxa and oftentimes disquisitional to survival. A common strategy is background matching, resembling the colour and pattern of the surroundings. This arroyo, even so, may be ineffective in complex habitats where matching i patch may atomic number 82 to increased visibility in other patches. In dissimilarity, confusing coloration, which disguises body outlines, may be effective confronting circuitous backgrounds. These ideas accept rarely been tested and previous work focuses on artificial systems. Here, nosotros test the camouflage strategies of the shore crab (Carcinus maenas) in ii habitats, being a species that is highly variable, capable of plastic changes in advent, and lives in multiple environments. Using predator (bird and fish) vision modelling and paradigm analysis, we quantified background matching and disruption in venereal from rock pools and mudflats, predicting that disruption would dominate in visually complex stone pools but background matching in more compatible mudflats. As expected, stone pool individuals had significantly college border disruption than mudflat crabs, whereas mudflat crabs more closely matched the substrate than rock pool crabs for colour, luminance, and pattern. Our study demonstrates facultative expression of camouflage strategies dependent on the visual environs, with implications for the evolution and interrelatedness of defensive strategies.
Introduction
Many animals exhibit visual similarities with their environment, commonly referred to as 'phenotype-environment associations'ane,2. It has long been appreciated that these associations are a product of natural option3,4, with individuals camouflaged confronting the prevailing visual environmentfive. Indeed, selection has driven correlations in advent between individual phenotypes and backgrounds in a wide range of animals, from rodents and lizards in terrestrial habitatshalf dozen,7 to crabs in marine habitats2,8,9, and even in plants10. These examples of phenotype-environment associations are highly suggestive of a camouflage function, simply past work has rarely demonstrated or quantified the actual camouflage resulting from whatsoever match to the local environment; that is, phenotype-environment matching (but see1). Nonetheless, camouflage, and its effectiveness, is widely appreciated to involve an interaction between the appearance of the organism and that of its groundwork. This poses the question of how camouflage should exist tuned to work best in different visual environments and contexts. Indeed, a great deal of work in bogus systems has explored how camouflage can be optimised under specific contexts (see below).
Phenotype-environment associations have to appointment largely been considered in the context of background matchingxi,12, a widespread class of camouflage involving resembling the general colour and design of the environment (e.g.13). Background matching reduces the deviation in features between the advent of an animal and its surroundings, and is therefore specific to the visual background where information technology has arisen12. While not always direct quantified in animals, background matching is probable to be widespread across many habitats and species, ranging from classic examples of concealment (due east.m.fourteen,xv) to studies demonstrating its utility in preventing detection of potential prey in both the lab (east.1000.16) and field13. At to the lowest degree one written report1 has also provided direct quantification of site-specific background matching in an intertidal crustacean, the sand flea (Hippa testudinaria), demonstrating that individuals match the color and luminance of their own beaches more than closely than of neighbouring beaches.
The efficacy of background matching tin can be limited by the outline of an fauna's torso, creating discontinuities with the background that make it more conspicuous to predators17. As a solution, confusing coloration involves relatively high contrast markings about the edge of the trunk to break up the outline3,17,18. Considerable research on disruptive coloration has demonstrated how this works, using bogus (man-made) casualty presented to either birds or humans (east.g.19,20,21,22,23,24). These studies accept shown that confusing coloration oft provides a reduction in detection (and potentially identification too), over and above the benefits conferred by background matching, and works at least in part by creating imitation edge data and hiding truthful torso outlines18,25.
Disruptive coloration has been suggested as a key method of camouflage across numerous taxonomic groups (see review26). However, studies have seldom quantified the camouflage effect of disruptive coloration in real animals, or even conspicuously demonstrated its presence. Work has been overwhelmingly focussed on bogus systems, or has largely subjectively inferred disruption. Merilaita27 analysed the distribution of spots on the polymorphic marine isopod Idotea baltica and constitute that spots deviated from a random distribution, being more likely to occur at the body edges. While consequent with disruption, this report did not compare the distribution of trunk markings to the actual distribution in the environment, nor directly quantify any potential disruptiveness of the markings either to vision models or in behavioural trials. A more contempo written report used a model of border disruption to assess the behavioural selection of resting position in ii species of moth, and found that both disruptive coloration and background matching were used to provide camouflage28. A similar earlier model also showed evidence for disruptive coloration and simulated edges in frogs29.
Overall, there remain several substantial gaps in our understanding of how camouflage is utilised in nature. Every bit discussed, much by work on phenotype-surroundings matching has relied on human subjective judgement and does not quantify actual camouflage match among habitats; groundwork matching is oftentimes inferred rather than direct quantified. 2d, electric current evidence for the utilise of disruption in nature remains very limited and seldom quantified in any existent animal. The vast majority of work is express to artificial (human being-fabricated) systems. Tertiary, no study has tested and compared the occurrence of both groundwork matching and confusing coloration strategies in an creature, especially in phenotypically variable species, nor investigated the use of these strategies across habitats. As nevertheless, few quantitative tests exist straight comparison how camouflage types are expected to vary in utilise with habitat type/spatial scene, and predictions from artificial systems require testing in real animals and environments. Finally, while both background matching and disruptive coloration are effective ways of preventing detection, in that location has been contend as to what extent these strategies operate independently21,xxx,31,32. Indeed, some studies suggest that maximum camouflage is achieved when both strategies are used in conjunction, and past work ofttimes shows that the benefits of disruption decline as body markings increasingly mismatch the backgroundxxx,32,33. However, once more these studies accept been restricted to simple bogus prey markings and few visual backgrounds (mostly tree bark).
A valuable group to exam the relative occurrence and tuning of camouflage strategies is crabs, which testify great diverseness in colours and patterns within and amid species, including for camouflage34,35. In venereal, phenotype-surround associations are thought to be common with many suggested examples, oft including species that can vary greatly in appearance2,8,9,36,37. Yet, these studies generally lack quantification of the match between individuals and the environment (but see38). Multiple studies accept involved the common shore crab (Carcinus maenas). Early descriptive work showed associations betwixt appearance and crab size/historic period and habitat39,40. More contempo work found similar results, including showing associations between crab colour patterns in dissimilar habitats (mussel beds, rock pools, seaweed, sandy beach and rocks) at varied spatial scales2,9,41, including when considering predator visioneight. In full general, crabs from mudflats tend to be more uniform, greener, and with less patterning than crabs from rock pools. In improver, more than uniform environments (e.chiliad. mudflats) harbour venereal with lower intraspecific diversity than heterogeneous habitats (eastward.g. rock pools)9. Mudflat crabs are thought to use habitat-specific background matching, whereas crabs from rock pools possess high dissimilarity and prominent markings, often found near the body edges, that are highly suggestive of disruptive colorationii.
In this study, we test for habitat-specific cover-up strategies in shore crabs across two habitat types. Using quantitative image assay and predator (bird and fish) vision modelling (Fig. 1), we compare the match of venereal to their backgrounds for color, luminance (perceived lightness), and pattern, and the extent of confusing coloration using recent metrics to appraise both pattern matching and border disruption that have been demonstrated to predict visual detection and predation rates13,23. We predict that shore crabs from heterogeneous and contrasting rock pool habitats cover-up themselves through disruptive mechanisms, whereas venereal from uniform mudflats employ groundwork matching in their low-contrast, homogenous environment.
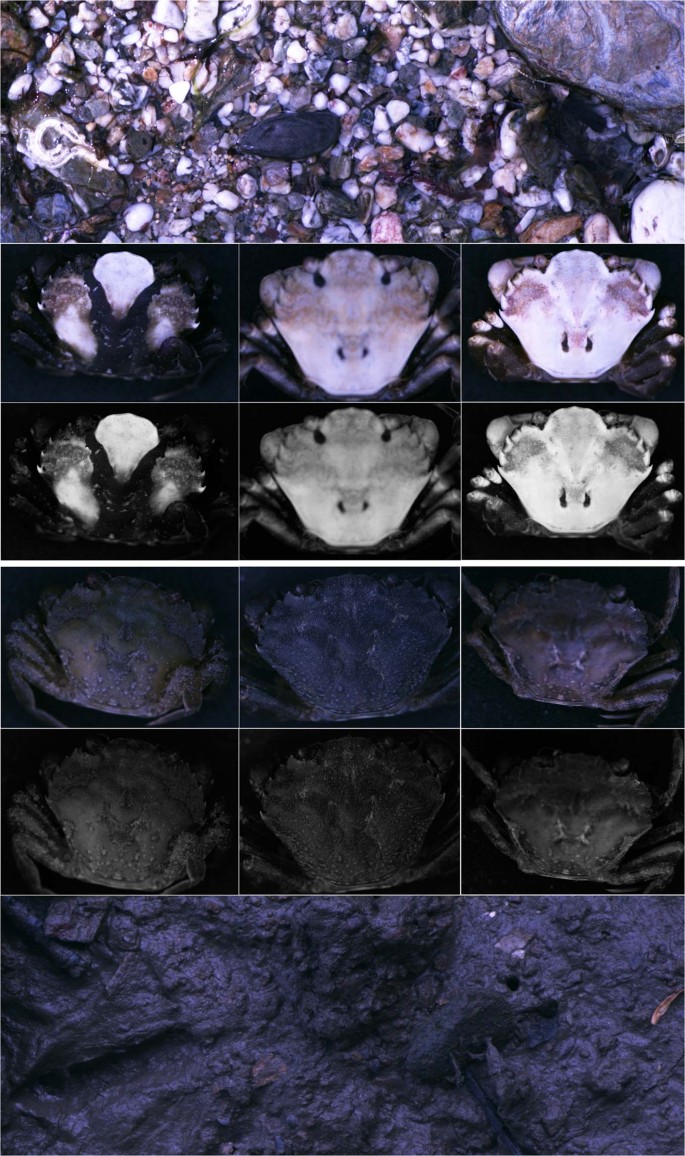
Images of a representative stone pool background (height; without crabs) and iii rock pool crabs with high confusing coloration scores. All images are converted to modelled avian vision. The peak row of crab images are colour, with the scarlet and greenish image layers comprising data from the LW and MW cones, and the blue layer comprising the combined information for the SW and UV cones (since images tin can merely display three colour layers). The second row of images are luminance (lightness) images corresponding to the double cones. The images of venereal below are for individuals from mudflat habitats (color and luminance), with an example mudflat background at the lesser.
Results
Groundwork matching: colour and luminance
To appraise how well crabs matched the colour and luminance of their backgrounds, bigotry or just noticeable deviation (JND) values were used to make up one's mind the departure between the carapace of an individual and the background (rock pool or mudflat). With the avian vision model, colour JND values were significantly predicted by an interaction betwixt crab habitat of origin and the groundwork habitat information technology was compared to (Fone,93 = 4.09, p = 0.045, Fig. 2). This indicates that the level of background matching for colour was dependent on the habitat the individual was collected from, just the magnitude of the upshot differed betwixt habitat types. The closest match to the groundwork (lowest JND) was seen in crabs collected from mudflats against mudflat habitat images (avian model average JND = one.65). Conversely, the poorest match was in crabs collected from stone pools confronting a rock pool background (avian average JND = 2.05). Rock pool individuals on mudflat backgrounds (avian average JND = 1.76) were a marginally better lucifer to the groundwork than rock pool venereal on a rock pool background. Using the fish vision model we found no significant interaction betwixt source habitat and background. The background main consequence was significant (F1,93 = half-dozen.11, p = 0.015). JND values were lower for fish vision relative to avian vision (meet Fig. 2). Overall, however, colour matches to each background type were shut (depression JNDs) for all color comparisons regardless of the specific combination of habitat type and crab habitat origin, for both avian and fish visual systems (JND average matches were all between 1.00 and two.x). Thus, color camouflage was proficient regardless of crab-habitat match.
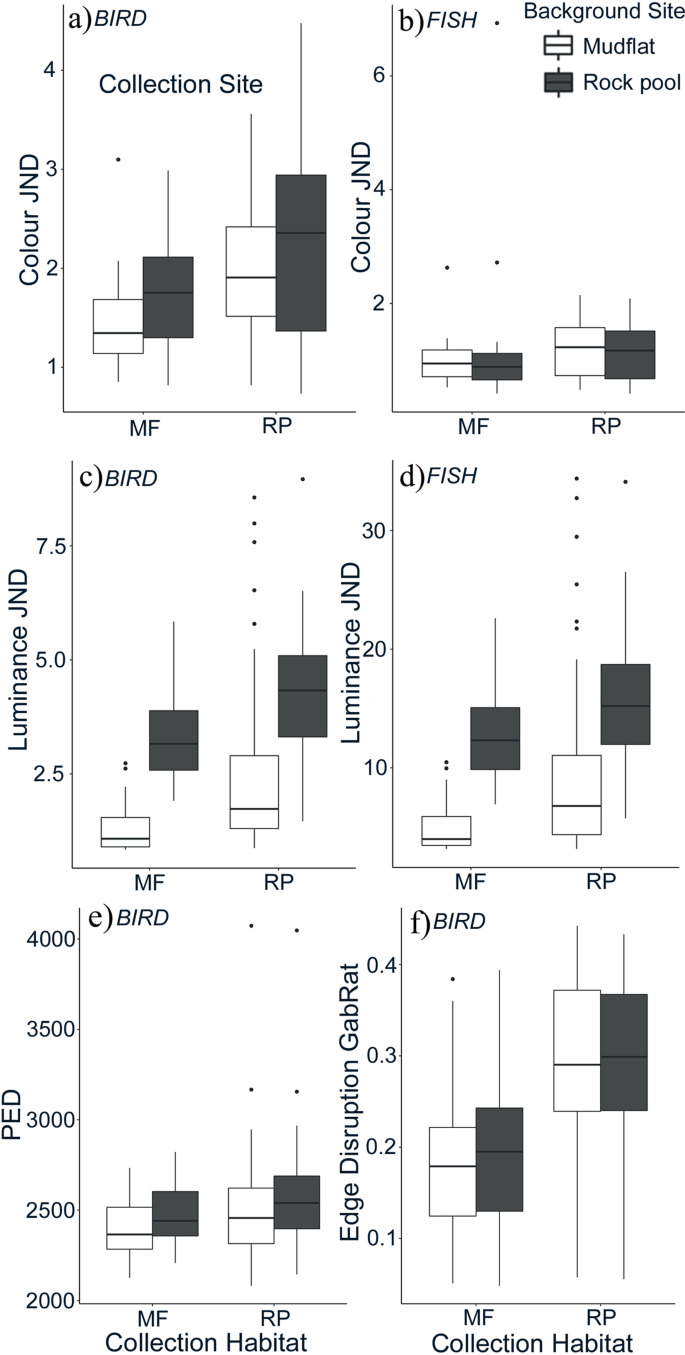
Discrimination values (JNDs) for venereal collected from either mudflat (MF) or rock puddle (RP) sites (Ten axis) compared to either mudflat or rock puddle backgrounds, corresponding to (a) avian color, (b) fish colour, (c) avian luminance, and (d) fish luminance vision comparisons. In all cases except (b) crabs are a closer match to mudflat than rock pool substrates, especially when originating from mudflat backgrounds. Plots (east and f) show results from pattern analyses for groundwork matching and disruptive coloration, respectively. For groundwork matching, smaller design free energy differences (PED) equate to a closer match. Hither, mudflat crabs show closer matches (better camouflage) than rock puddle venereal to the mudflat substrate. For disruption, larger scores (GabRat) equate to higher disruptive coloration, and here stone pool crabs evidence greater disruptive coloration than mudflat crabs. Boxplots show boilerplate values (bold line), interquartile range (box component), range of minimum and maximum values, and outliers (circles).
Luminance JND betwixt crab carapace and background was analysed using both avian and fish visual systems. In that location was a pregnant interaction betwixt the crab habitat of origin and the groundwork type for both visual systems (avian: F1,93 = 10.98, p = 0.001; fish: F1,93 = viii.03, p = 0.006, Fig. 2). Similar to the colour JND, the lowest luminance JND values came from shore venereal collected from mudflat sites against a mudflat groundwork. Differences were more marked for luminance than for the color comparisons. For avian vision, average matches of crabs to mudflat habitats were one.79 and 1.95 JNDs for crabs from mudflats and stone pools respectively. That is compared to analogous matches of three.59 and 3.73 JNDs for crabs against rock pool backgrounds. For fish vision, luminance average JNDs for crabs against mudflats were 6.76 and 7.35, versus matches of thirteen.40 and xiii.92 for crabs against rock pools.
Background matching: design
To assess how well venereal matched the pattern of their backgrounds, pattern energy difference (PED) values were used to determine the divergence in design spectra between the carapace of an individual and the background habitat (rock puddle or mudflat). In that location was a pregnant interaction between crab habitat and background habitat (F1,88 = 4.66, p = 0.033; Fig. 2). This indicates that the level of pattern background matching depended on the habitat the individual was collected from, but the magnitude of the effect differed between habitat types. The closest match to the background (lowest PED) was seen in venereal collected from mudflats and against mudflat habitat images. Conversely, the largest difference between carapace and background was in crabs nerveless from rock pools against a rock puddle background. Otherwise, rock pool individuals on mudflat backgrounds had lower PED values and therefore were a better match to the background than rock pool crabs on a rock pool background. This shows that the phenotype of rock puddle venereal is actually closer in design to the backdrop of mudflats than rock pools.
Disruption
GabRat measurements of crab carapace disruption produced values ranging from very depression (0.04) to values considered highly disruptive (>0.40)23. At that place was a meaning interaction between the habitat venereal originated from and the background habitat they were compared to (interaction: Fane,84 = 9.96; P = 0.002, Fig. 2). Overall, crabs nerveless from rock pools had significantly higher levels of border disruption against both rock pool and mudflat groundwork than crabs from mudflats. The edge disruption of venereal collected from rock pool sites was not greatly unlike against rock pool every bit opposed to mudflat backgrounds, although rock pool crabs had marginally more disruptive edges on mudflat backgrounds than they did confronting their own habitat substrate (Fig. 2). Conversely, mudflat crabs have slightly higher edge disruption levels on rock pools than against their own substrate background.
Discussion
We assessed 2 unlike cover-up strategies (background matching and disruptive patterning) in juvenile shore crabs collected from rock pool and mudflat habitats. In line with our predictions, there was a clear difference in the caste of similarity of crabs to the background environment and in levels of border disruption, depending on whether crabs were collected from rock pools or mudflats. Specifically, crabs collected from the more heterogeneous rock pools had a college level of disruptive patterning than crabs from mudflats, and this issue was in fact marginally more pronounced against a mudflat background than confronting a rock pool background. This indicates that the disruptive outcome operated on both backgrounds and is therefore somewhat intrinsic to the crabs themselves. Analysis of background matching found that crabs nerveless from the more homogeneous mudflat habitats were better matched to mudflat backgrounds than to stone puddle backgrounds, and this was mostly the instance for colour, luminance, and blueprint. Rock puddle venereal compared to rock puddle backgrounds often had the lowest level of background matching, whereas mudflat crabs against mudflat backgrounds had the closest overall matches. However, nosotros notation that for colour the visual modelling indicates that the level of match, and therefore camouflage, was very good for all color comparisons regardless of habitat. Consequent with this, for colour, there was footling difference in matching to a fish visual system, and only a marginally better friction match of venereal to the mudflat background to the avian model. In contrast, the results for luminance lucifer were clearer than for colour, with mudflat crabs a closer match to their habitat for both visual systems. This was especially the case for the fish visual model. Overall, rock puddle individuals match their habitat less closely just accept high levels of edge disruption, whereas mudflat individuals do not accept disruptive markings but match their background closely for color, design, and luminance. These findings support our predictions that differences in cover-up strategy arise between individuals institute in mudflat and stone pool habitats.
Our results build on earlier piece of work investigating shore crab appearance, which demonstrated phenotype-surroundings associations betwixt carapace patterns and habitat at multiple spatial scales2,eight,nine,41. Previous piece of work likewise showed that venereal from more uniform habitats exhibited less carapace patterning than crabs from heterogeneous habitats. The prominent markings establish on some venereal, which tend to arise at the trunk margins, have also been suggested to provide disruptive camouflage2. Our written report also ties in with other recent work, showing that with ontogeny, crabs tend to adopt more than uniform night-dark-green appearances, closer to mudflat backgrounds, and that this translates into a survival reward42. Overall, shore crabs therefore appear to utilise strategies that are well suited to the environments in which they live. Side by side, experiments are needed to test the effectiveness of disruptive edges on the survival (or predator detection) of shore crabs. This is non-lilliputian, all the same, since the changing nature of the environs (due to tides, waves, weather) makes experiments based on model prey (e.g. made of Plasticine) challenging to design robust yet visually accurate prey and record predation events, as well as targeting the wide diversity of predatory taxa.
Our piece of work demonstrates how the presence and likely utility of different cover-up markings depends on the visual environment. While this is often discussed12,18,43, little work has tested such ideas, especially in natural systems. In compatible environments, a close match to large areas of the groundwork is possible since in that location is as well no ambiguity over the best colour or pattern to lucifer. In low dissimilarity changeless habitats, disruption involving high dissimilarity non-matching markings may actually give an private away. In contrast, in highly variable backgrounds while it may be possible for different individual phenotypes to match dissimilar patches44, or for animals to prefer generalist or 'compromise' camouflage strategies that match no background perfectly but several to some caste45,46, it may sometimes still be better to match the about common substrate47. Alternatively, as we demonstrate here, some other approach is to focus on using disruptive camouflage, which may provide a ameliorate route to concealment irrespective of the specific groundwork patches encountered18,26. In addition, the visually complex nature of the stone pool environment probable offers further protection independent of groundwork matching, since high background complication is known to facilitate camouflage48,49.
Disruption is widely considered to work best aslope groundwork matching30,32, though there is evidence that to an extent it may likewise work independently and permit camouflage fifty-fifty with relatively mismatched individuals21. Here, we accept shown in a natural system, with complex real creature markings and ii different background types, that disruption seems to be utilised when background matching is ineffective, and vice versa. Why rock pool crabs had marginally stronger disruptive effects when superimposed onto mudflat backgrounds is unclear, but a possible caption may be that rock pool venereal seen confronting homogeneous habitats benefitted from higher levels of differential blending, enabling large parts of the carapace to blend into the background while other aspects stood out3,xviii. Regardless, this finding provides support that disruptive patterning is a distinct camouflage strategy not fully reliant on matching the background22.
While rock pool crabs have greater levels of disruption, this may not be the sole utility of their patterns. Various markings may contribute to groundwork matching in some instances, and loftier levels of phenotypic diverseness (peculiarly in rock pool habitatsix) may reduce the ability of predators to detect individuals past hindering the formation of search images50. Furthermore, in common with other crustaceans, carapace patterns in shore crab go less singled-out with age8,9,37,39,42,51. Hither, we compared crabs of the same size range and so a reduction in pattern in mudflat crabs cannot exist explained by ontogenetic changes. An absence of pattern could in theory also be caused by strong visual predation removing patterned venereal from mudflat environments. Yet, predation levels are unlikely to be high enough for this, or to override the strong recruitment of new individuals into each location via continuous settlement of post-larvae from their planktonic larval stage and through motion of crabs8,9,41. More likely, our results stem from the considerable plasticity in individual shore crab blueprint and coloration35,twoscore,52. An additional possibility is that individuals show groundwork choice of substrates for concealment, which has been found in a range of other species and taxa53, including ghost crabs54. Indeed, microhabitat associations in shore crab appearance and substrate have been found in past work that are difficult to explain without some level of background choice existing8,41. However, by piece of work direct investigating background choice has focussed on decisions inside habitats and oft at very fine spatial scales, and here we are considering sites and habitats separated past several km. While shore crabs accept been known to move up to 2 km in a short infinite of fourth dimension55, it seems implausible that they move such distances and among habitats for the purpose of selecting appropriate background environments for camouflage. However, this remains an area that would benefit from farther enquiry.
Our work highlights the need to understand camouflage strategies more comprehensively in natural systems. A range of previous studies on beast coloration take undertaken comparative analyses, finding testify for links between camouflage and other defensive strategies with aspects of habitat and life-history (e.grand.56,57). Disruptive coloration has been suggested to be extremely common in nature26, just it is seldom quantified, nor how its characteristics relate to the visual surround in which the bearer is found. Other work, especially on rapid colour irresolute (in seconds) species such as cuttlefish has explored the expression of camouflage patterns on different visual backgrounds, showing for example that backgrounds of different contrasts and spatial data tend to elicit more than uniform or pronounced marking types31. Ane study in particular has also investigated both color and pattern match of color-changing flatfish to a range of visual systems, showing that expression of markings is habitat-dependent and that the flounder lucifer the spatial scale of sand and gravel well, but not rocks58. This latter finding is interesting considering it is consistent with the crabs in our study showing poor match to the stone pool substrates. All the same, the above study deviates from ours in that information technology analysed groundwork matching only not disruptive coloration, and did not exam how cover-up types/strategies are expected to vary in use with habitat type. In general, more than piece of work is needed in this area.
Finally, another key area for further investigation is how phenotype-environment matching links to non-reversible ontogenetic changes in advent with age and size. In shore crabs, we have too shown that larger venereal progress to a more uniform green coloration as they grow, at to the lowest degree partly independently from the groundwork environs, and that this appears to facilitate generalist camouflage beyond many habitats, especially when crabs are larger and more mobile42. To what extent ontogenetic and plastic changes explain overall phenotypes, and how this relates to camouflage strategy, requires considerable hereafter work. Given that the expression and optimisation of cover-up has been a fruitful surface area for exploring a variety of issues, from evolutionary outcomes and mechanisms of adaptation (east.g.59) through to how visual perception worksthreescore, testing the presence and efficacy of camouflage features in circuitous visual scenes in existent animals is an important surface area. This too has the potential to inform on how conspicuous signals improve efficacy in different habitats, from sexual and social signals to aposematism.
Methods
Field Sites and Photography
Images were taken of rock pool and mudflat habitats across six sites, iii were stone pool habitats (47 background images) and 3 were mudflats (47 images). Although there was some variation in features amidst rock puddle sites, in general the groundwork substrate was similar, consisting of large clusters of rocks, forming deep gullies filled with minor pebbles and sand, alongside pocket-sized pools (encounter9 for habitat assessments; Fig. 1). Conversely, mudflats consist of large expanses of dark brown mud and surface algae, with picayune shelter other than dispersed rocks or objects. Sites were located on both north and south coasts of Cornwall, Southwest United kingdom of great britain and northern ireland and were separated by between six and 50 km. Gyllyngvase beach (fifty° 8′ 39.42″North, −5° 4′ 5.244″W) in the Falmouth area, Kennack Sands (50° 0′ 23.695″North, −v° 9′ 28.258″West) located further downwards the southwest coast, and Perranuthnoe (fifty° 6′ 43.383″Northward, −5° 26′ 28.142″West) on the south coast were rock pool sites. For mudflats, Penryn (l° 9′ 49.335″North, −5° 5′ 2.124″W) and Helford (l° 5′ 23.i″N, −5° nine′ 58.754″W) were chosen on the southward declension, with Hayle (50° 11′ 36.979″Due north, −5° 25′ 47.973″W) on the north coast. After entry to the focal surface area of a site, images were taken separated by approximately 3.7 thou with 20 images taken at each site. Some images were deselected back in the lab if they were deemed to be also out of focus, resulting in 47 images overall per habitat. All work was conducted under blessing from the University of Exeter ideals committee (application number: 2016/1162).
Image conquering followed standard protocolseight,9,thirteen. Images of backgrounds were taken with a Nikon D7000 digital camera modified with a quartz conversion to let for UV sensitivity (Avant-garde Camera Services, Norfolk, Great britain) fitted with a Nikon Nikor 105 mm lens. All photographs were taken in RAW format with fixed discontinuity settings. The camera was held in position using a tripod and all photographs were taken at the aforementioned height (approximately 1 m). Two sets of images were taken, using a visible (Baader UV/IR Cut filter) and UV (Baader Venus U filter) filter, to cake UV and infrared lite (human being-visible images) and allow only UV transmission between 300–400 nm (UV images), respectively. Our photographic camera sensitivities are as follows: UV: 360–400 nm (peak 380 nm), SW: 400–550 nm (acme 460 nm), MW: 420–620 nm (peak 540 nm), LW: 560– 700 nm (peak 625 nm)61. To go along calorie-free conditions compatible, images were taken on clouded, cloudy days. A photographic umbrella was likewise used for each photo to minimise glare, and a black and white reflectance standard with a scale bar was placed in the corner of each image for subsequent image calibration and standardisation62. The standard was fabricated from 10 × 10 mm sections of zenith diffuse sintered PTFE sheet (Labsphere, Congleton, United kingdom of great britain and northern ireland) and reflected eight.2% and 94.8% of all wavelengths respectively63. A scale bar was used to automatically resize all images to the same scale for subsequent design and disruption analysis.
For the purposes of this study, nosotros focussed on small-scale juvenile crabs with <15 mm carapace width (CW) at the widest point. While past work has categorised venereal as 'adults' when CW > 25 mm, there is a gradual decrease in patterning and a modify in the body appearance towards more uniform green as crabs develop2,8,ix. The reasons for this likely reflect a switch to a generalist camouflage strategy in adults that are more mobile across sites than juveniles58. We therefore focus on modest juvenile crabs that are much more than likely to require habitat-specific cover-up and which are likely to incur greater predation adventure than adults8,34,63. In full, 97 venereal were sampled and used for the groundwork matching analyses (colour, luminance, and pattern) and 86 of these were used for the disruptive coloration analysis.
Collection of crabs followed by approaches8,9,52,58, with sampling at depression tide by systematically searching the substrate, lifting seaweed, rocks, and raking the substrate with fingers to locate individuals in a given area. Crabs were then transported back to the laboratory at the Academy of Exeter, Penryn Campus in clear tanks containing table salt h2o from the habitat and background substrate to cover the bottom of the tank, providing refuge to avoid inflicting stress during transportation. Individuals were then gently dried with tissue newspaper and placed underneath a tripod set in a dark photography room. Each crab was placed on a spectrally flat sail of 2 mm thick black foam with a reflective white PTFE cylinder surrounding the individual to diffuse the light for photography.
Image assay and vision modelling
Multispectral images were created using the 'multispectral epitome calibration and analysis toolbox' in Epitome J61. Images were aligned and the white and black standards selected to allow images to be linearised with regards to radiance and standardised to control for light conditions61,62. Images were resized downwards to the same scale using the scale bar9. Once these images had been calibrated, regions of interest (ROIs) were selected for measurement. Here, the carapace of each crab, excluding appendages, was selected.
We modelled the visual organization of both a predatory bird and fish. Among the chief predators of shore crabs are shore birds and fish37,64. Most birds are probably tetrachromats, using four cone types in colour vision: longwave (LW), mediumwave (MW), shortwave (SW) and ultraviolet/violet (UV/V). Most shore birds have a 'violet' cone type relatively more than sensitive to longer violet wavelengths than some other more UV-sensitive birds65. Therefore, for modelling nosotros followed8 and used the visual sensitivity of the peafowl (Pavo cristatus)66, which is widely used as a species for visual modelling of violet birds (Fig. 1). Although other birds that may exist relevant predators (e.g. gulls) can accept an 'ultraviolet' blazon arrangement more sensitive to UV light, the crabs and backgrounds in our report generally have low ultraviolet reflectance. For fish, the European pollack, Pollachius pollachius 67 is thought to be a fundamental predator of crabs and represents a dichromatic fish predator, with LW and SW sensitive cones. While fish in our study site can also be trichromats, past work has showed simply minor differences in modelled crab advent amongst di- and tetrachromatic systems8, and and so other visual systems are unlikely to vary greatly either. Nosotros converted standardized images to predicted cone catch information for each species using a widely implemented polynomial mapping technique62. This has been repeatedly shown to provide highly authentic data compared to cone catch modelling with reflectance spectra (see25,61,68).
Background matching: colour and luminance
To quantify colour and luminance match to the background we used a widely employed log version of a model of predator discrimination69. This calculates only noticeable differences (JNDs) between two objects to determine discriminability. The output of the model, JNDs, predicts whether ii objects can be discriminated (values < 1.00), with increasing values equating to a reduction in the level of camouflage match. For full details see Supplementary Fabric. We measured the cone values for each crab ROI for each visual system, then the aforementioned for each background image. Using the in a higher place models, nosotros then compared the colour and luminance match of each crab carapace to every background epitome, followed by calculating average colour and luminance JNDs for each crab to each habitat. Nosotros therefore derived an boilerplate level of background matching for each crab to each of the ii habitat types, across all samples.
Background matching: pattern
To assess background design matching between crab carapace and the groundwork for each habitat, a granularity analysis was conducted9,70,71 – see Supplementary Material. We used a modification of this procedure to make directly comparisons between the body markings of an animate being and the substrate ('pattern energy deviation', PED), giving a measure of background pattern matching that predicts detection by wild predators13 and humans searching for computer targets23. For pattern assay nosotros used the double cone (luminance) values of the peafowl (Pavo cristatus)66. Any two patterns with similar energy across all spatial scales will produce low pattern deviation values, indicative of background matching, whereas difference in either aamplitude or shape of the spectra will produce larger differences. Here, the absolute departure between the spectra of crab carapaces and habitat backgrounds was assessed (both rock pool and mudflat separately). As above, we derived an average level of pattern matching for each crab to each of the ii habitat types.
Disruptive coloration
To quantify edge disruption, nosotros used a recently adult method called 'GabRat', which uses angle sensitive filters to measure the ratio of false edges to coherent edges effectually the target outline23 – see Supplementary Cloth. A high ratio of fake edges to coherent edges should exist more disruptive, and therefore indicates that prey are more difficult to detect, while lower values suggest salient coherent edges. While we note that GabRat is relatively new and awaits greater testing, especially in natural systems, the metric has been shown to exist one of the near important predictors of human detection times of disruptive targets (and superior to other pattern metrics, including those for quantifying disruption based on more than conventional edge detection algorithms23). For each paradigm, each crab was randomly placed in fifty different positions that did not overlap with each other or whatsoever exclusion zones. This was repeated on all 94 backgrounds (47 rock pool and 47 mudflat), resulting in a full of 4700 edge disruption measurements per private crab. This process accounted for variation in positioning of crabs in the wild. The average GabRat value of the total 50 positions was calculated for each background, then that one value was generated per crab/image combination. Means per individual were and so calculated across both rock pool and mudflat backgrounds, and so that each crab had an boilerplate border disruption value for both habitat types.
Statistics
All individuals (from the ii habitat types) were placed onto both habitat types, resulting in 2 mean values per individual. A split plot 2 × 2 repeated measures mixed factorial ANOVA with blazon III sums of squares was used to appraise the match of individuals to stone pool and mudflat images using the R function ezANOVA. Our inside subjects gene was image background and the between subjects cistron was the collection habitat. Full models including the 2-way interaction were run for each of our dependent variables (the metrics of cover-up): border disruption information, PED, and colour and luminance data for both avian and fish vision models; and so the general grade was: (camouflage metric ~drove.site +groundwork.habitat +collection.site *background.habitat).
Homogeneity of variance was assessed using Levene'southward test. Colour JND data for avian vision was non-normal and so a log transformation was applied. Assumptions of normality and homogeneity of variance were met in all analyses, which were conducted in the statistical plan R72.
We predict that crabs from stone pool habitat sites volition take higher GabRat edge disruption values than venereal from mudflat habitats. There should besides be a habitat effect, with the variation in rock pool backgrounds allowing greater disruption regardless of the crab origins. Conversely, for background matching, venereal from mudflat habitats may ameliorate match the background for each metric owing to its more elementary nature than crabs from rock pool habitats. In improver, mudflat backgrounds may also allow greater background matching owing to their more uniform appearance, whereas rock pools present highly variable environments meaning matching many patches is not possible.
Data Availability
All data for this study are included every bit a Supplementary File.
References
-
Stevens, M. et al. Phenotype-surroundings matching in sand fleas. Biol Lett 11, 20150494 (2015).
-
Todd, P. A., Briers, R. A., Ladle, R. J. & Middleton, F. Phenotype-environment matching in the shore crab (Carcinus maenas). Mar Biol 148, 1357–1367 (2006).
-
Cott, H. B. Adaptive Coloration in Animals. (Methuen & Co. Ltd, 1940).
-
Wallace, A. R. Mimicry and other protective resemblances among animals. Westminster Rev (London ed.) one(July), one–43 (1867).
-
Stevens, M. & Merilaita, Southward. Introduction. Creature camouflage: current issues and new perspectives. Phil Trans R Soc B 364, 423–427 (2009).
-
Rosenblum, Eastward. B. Convergent evolution and divergent selection: Lizards at the White Sands ecotone. Am Nat 167, i–15 (2006).
-
Boratyński, Z. et al. Repeated evolution of camouflage in speciose desert rodents. Sci Rep 7, 3522 (2017).
-
Nokelainen, O., Hubbard, N., Lown, A. E., Woods, L. Due east. & Stevens, M. Through predators' optics – phenotype-environment associations in shore crab coloration at different spatial scales. Biol J Linn Soc 122, 738–751 (2017).
-
Stevens, M., Wood, Fifty. E. & Lown, A. E. Camouflage and individual variation in shore venereal (Carcinus maenas) from dissimilar habitats. PLoS One 9, e115586 (2014).
-
Niu, Y., Lord's day, H. & Stevens, Chiliad. Plant cover-up: ecology, evolution, and implications. TREE 33, 608–618 (2018).
-
Endler, J. A. Progressive background matching in moths, and a quantitative measure of crypsis. Biol J Linn Soc 22, 187–231 (1984).
-
Merilaita, Due south. & Stevens, M. In Animal Camouflage: Mechanisms & Office (eds Stevens, M. & Merilaita, S.) 17–33 (Cambridge University Press, 2011).
-
Troscianko, J., Wilson-Aggarwal, J., Stevens, Thousand. & Spottiswoode, C. N. Camouflage predicts survival in ground-nesting birds. Sci Rep 6, 19966 (2016).
-
Kettlewell, H. B. D. Selection experiments on industrial melanism in the Lepidoptera. Heredity ix, 323–342 (1955).
-
Walton, O. C. & Stevens, M. Avian vision models and field experiments determine the survival value of peppered moth cover-up. Comm Biol 1, 118 (2018).
-
Pietrewicz, A. T. & Kamil, A. C. Visual detection of cryptic prey by blue jays (Cyanocitta cristata). Science 195, 580–582 (1977).
-
Thayer, G. H. Concealing-Coloration in the Animal Kingdom: An Exposition of the Laws of Disguise Through Color and Blueprint: Beingness a Summary of Abbott H. Thayer's Discoveries. (Macmillan, 1909).
-
Stevens, M. & Merilaita, S. Defining confusing coloration and distinguishing its functions. Phil Trans R Soc B 364, 481–488 (2009).
-
Cuthill, I. C. et al. Confusing coloration and background pattern matching. Nature 434, 72–74 (2005).
-
Merilaita, S. & Lind, J. Background-matching and disruptive coloration, and the evolution of cryptic coloration. Proc R Soc B 272, 665–670 (2005).
-
Schaefer, M. H. & Stobbe, North. Disruptive coloration provides camouflage independent of background matching. Proc R Soc B 273, 2427–2432 (2006).
-
Webster, R. J., Hassall, C., Herdman, C. One thousand. & Sherratt, T. N. Confusing cover-up impairs object recognition. Biol Lett 9, 20130501 (2013).
-
Troscianko, J., Skelhorn, J. & Stevens, M. Quantifying camouflage: how to predict detectability from appearance. BMC Evol Biol 17, 7 (2017).
-
Sharman, R. J., Moncrieff, S. J. & Lovell, P. Thousand. Dissociating the effect of disruptive colouration on localisation and identification of inconspicuous targets. Sci Rep 8, 6599 (2018).
-
Stevens, M. & Cuthill, I. C. Confusing coloration, crypsis and border detection in early visual processing. Proc R Soc B 273, 2141–2147 (2006).
-
Stevens, M., Cuthill, I. C., Párraga, A. C. & Troscianko, T. In Progress in Encephalon Research Vol. 155 (eds Alonso, J.-M. et al.) 49–65 (Elsevier, 2006).
-
Merilaita, S. Crypsis through disruptive coloration in an isopod. Proc R Soc B 265, 1059–1064 (1998).
-
Kang, C. K., Stevens, M., Moon, J. Y., Lee, S. I. & Jablonski, P. G. Camouflage through behavior in moths: the role of background matching and disruptive coloration. Behav Ecol 26, 45–54 (2015).
-
Osorio, D. & Srinivasan, Yard. 5. Camouflage past edge enhancement in animal coloration patterns and its implications for visual mechanisms. Proc R Soc B 244, 81–85 (1991).
-
Fraser, S., Callahan, A., Klassen, D. & Sherratt, T. N. Empirical tests of the role of disruptive coloration in reducing detectability. Proc R Soc B 274, 1325–1331 (2007).
-
Hanlon, R. T. et al. Cephalopod dynamic camouflage: bridging the continuum between background matching and disruptive coloration. Phil Trans R Soc B 364, 429–437 (2009).
-
Stevens, M., Cuthill, I. C., Windsor, A. Chiliad. M. & Walker, H. J. Disruptive dissimilarity in animal camouflage. Proc R Soc B 273, 2433–2438 (2006).
-
Stobbe, North. & Schaefer, M. H. Enhancement of chromatic contrast increases predation adventure for striped collywobbles. Proc R Soc B 275, 1535–1541 (2008).
-
Caro, T. Yard. The functional significance of coloration in crabs. Biol J Linn Soc 124, 1–10 (2018).
-
Stevens, M. Color modify, phenotypic plasticity, and camouflage. Front end Ecol Evol 4, 51 (2016).
-
Jensen, M. C. & Egnotovich, M. Due south. A whiter shade of male person: color groundwork matching as a function of size and sex in the yellow shore crab Hemigrapsus oregonensis (Dana, 1851). Curr Zool 61, 729–738 (2015).
-
Palma, A. T. & Steneck, R. T. Does variable coloration in juvenile marine crabs reduce risk of visual predation? Ecology 82, 2961–2967 (2001).
-
Russell, B. J. & Dierssen, H. Grand. Use of hyperspectral imagery to assess cryptic color matching in Sargassum associated crabs. PLoS One 10, e0136260 (2015).
-
Hogarth, P. J. Variation in the carapace design of juvenile Carcinus maenas. Mar Biol 44, 337–343 (1978).
-
Powell, B. L. The responses of the chromatophores of Carcinus maenas (L. 1758) to light and temperature. Crustaceana four, 93–102 (1962).
-
Todd, P. A., Oh, J., Loke, Fifty. H. L. & Ladle, R. J. Multi-scale phenotype-substrate matching: Evidence from shore venereal (Carcinus maenas L.). Ecol Circuitous 12, 58–62 (2012).
-
Nokelainen, O., Maynes, R., Mynott, Due south., Toll, N. & Stevens, M. Improved camouflage through ontogenetic color change confers reduced detection adventure in shore crabs. Func Ecol 33, 654–669 (2019).
-
Merilaita, S., Scott-Samuel, N. E. & Cuthill, I. C. How camouflage works. Phil Trans R Soc B 372, 20160341 (2017).
-
Endler, J. A. A predator'due south view of creature color patterns. Evol Biol 11, 319–364 (1978).
-
Houston, A. I., Stevens, M. & Cuthill, I. C. Animate being camouflage: compromise or specialise in a 2 patch-blazon environs? Behav Ecol eighteen, 769–775 (2007).
-
Merilaita, S., Tuomi, J. & Jormalainen, V. Optimization of cryptic coloration in heterogeneous habitats. Biol J Linn Soc 67, 151–161 (1999).
-
Michalis, C., Scott-Samuel, N. E., Gibson, D. P. & Cuthill, I. C. Optimal background matching camouflage. Proc R Soc B 284, 20170709 (2017).
-
Merilaita, S. Visual background complexity facilitates the evolution of camouflage. Development 57, 1248–1254 (2003).
-
Xiao, F. & Cuthill, I. C. Background complication and the detectability of camouflaged targets past birds and humans. Proc R Soc B 283, 20161527 (2016).
-
Bond, A. B. & Kamil, A. C. Visual predators select for crypticity and polymorphism in virtual prey. Nature 415, 609–613 (2002).
-
Todd, P. A., Qiu, West. & Chong, K. Y. Ontogenetic shifts in carapace patterning and/or colouration in intertidal and subtidal brachyuran crabs. Raffles Balderdash Zool 57, 543–550 (2009).
-
Stevens, M., Lown, A. Eastward. & Woods, L. Due east. Color change and camouflage in juvenile shore venereal Carcinus maenas. Front Ecol Evol two, xiv (2014).
-
Stevens, One thousand. & Ruxton, M. D. The fundamental role of behaviour in beast cover-up. Biol Rev94, 116–134 (2019).
-
Uy, F. G. K. et al. Active groundwork choice facilitates crypsis in a tropical crab. Biotropica 49, 365–371 (2017).
-
Ameyaw-Akumfi, A. & Naylor, E. Spontaneous and induced components of salinity preference behaviour in Carcinus maenas. Mar Ecol Prog Ser 37, 153–158 (1987).
-
Allen, Due west. Fifty., Baddeley, R., Scott-Samuel, N. East. & Cuthill, I. C. The evolution and function of pattern diverseness in snakes. Behav Ecol 24, 1237–1250 (2013).
-
Stoner, C. J., Caro, T. M. & Graham, C. M. Ecological and behavioral correlates of coloration in artiodactyls: systematic analyses of conventional hypotheses. Behav Ecol xiv, 823–840 (2003).
-
Akkaynak, D., Siemann, 50. A., Barbosa, A. & Mäthger, L. M. Changeable camouflage: how well tin flounder resemble the colour and spatial calibration of substrates in their natural habitats? R. Soc. Open Sci. four, 160824 (2017).
-
Nachman, M. W., Hoekstra, H. Due east. & D'Agostino, Due south. L. The genetic basis of adaptive melanism in pocket mice. PNAS 100, 5268–5273 (2003).
-
Troscianko, T. S., Benton, C. P., Lovell, P. Thou., Tolhurst, D. J. & Pizlo, Z. Camouflage and visual perception. Phil Trans R Soc B 364, 449–461 (2009).
-
Troscianko, J. & Stevens, M. Image calibration and assay toolbox – a gratis software suite for objectively measuring reflectance, colour and pattern. Meth Ecol Evol half dozen, 1320–1331 (2015).
-
Stevens, Yard., Párraga, C. A., Cuthill, I. C., Partridge, J. C. & Troscianko, T. S. Using digital photography to report animal coloration. Biol J Linn Soc 90, 211–237 (2007).
-
Crothers, J. H. The biology of the shore crab Carcinus maenas (L.): The background anatomy, growth and life history. Field Stud 2, 407–434 (1967).
-
Crothers, J. H. The biology of the shore crab Carcinus maenas (L.): The life of the adult crab. Field Studies 2, 579–614 (1968).
-
Ödeen, A., Håstad, O. & Alstrom, P. Evolution of ultraviolet vision in shorebirds (Charadriiformes). Biol Lett 6, 370–374 (2010).
-
Hart, N. Due south. Vision in the peafowl (Aves: Pavo cristatus). J Exp Biol 205, 3925–3935 (2002).
-
Shand, J., Partridge, J. C., Acher, Southward. North., Potts, Yard. W. & Lythgoe, J. N. Spectral absorbance changes in the violet/blue sensitive cones of the juvenile pollack, Pollachius pollachius. J Comp Physiol A 163, 699–703 (1988).
-
Expressway, T. West. Using digital cameras to investigate animal colouration: estimating sensor sensitivity functions. Behav Ecol Sociobiol 65, 849–858 (2011).
-
Vorobyev, M. & Osorio, D. Receptor noise as a determinant of color thresholds. Proc R Soc B 265, 351–358 (1998).
-
Chiao, C.-C., Chubb, C., Buresch, K. C., Siemann, L. & Hanlon, R. T. The scaling effects of substrate texture on camouflage patterning in cuttlefish. Vis Res 49, 1647–1656 (2009).
-
Stoddard, Grand. C. & Stevens, G. Pattern mimicry of host eggs by the mutual cuckoo, as seen through a bird'south eye. Proc R Soc B 277, 1387–1393 (2010).
-
Team, R. C. R: A Language and Surroundings for Statistical Computing. (R Foundation for Statistical Computing, Vienna, Austria, 2013).
Acknowledgements
We give thanks Tim Caro and Neeltje Boogert for comments on an earlier version of this piece of work. This work was partly supported by a BBSRC grant (BB/L017709/ane) to MS.
Author data
Authors and Affiliations
Contributions
N.P., Chiliad.Due south. and T.T. designed and the study. Northward.P. conducted fieldwork and data collection, and analysed the results with input from Yard.S. and T.T. S.Chiliad. undertook the colour and luminance image analysis work. J.T. helped North.P. with confusing coloration image analysis. M.Due south. and N.P. wrote the manuscript with input from all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits employ, sharing, adaptation, distribution and reproduction in any medium or format, as long equally you give appropriate credit to the original writer(s) and the source, provide a link to the Creative Eatables license, and indicate if changes were made. The images or other 3rd political party material in this article are included in the commodity's Artistic Eatables license, unless indicated otherwise in a credit line to the cloth. If material is not included in the commodity's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will demand to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
Nigh this article
Cite this article
Cost, N., Green, South., Troscianko, J. et al. Background matching and confusing coloration equally habitat-specific strategies for camouflage. Sci Rep ix, 7840 (2019). https://doi.org/10.1038/s41598-019-44349-ii
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/s41598-019-44349-2
Further reading
-
Spatio-temporal color variation of arthropods and their surround
Evolutionary Ecology (2022)
-
Biomimetic chameleon soft robot with artificial crypsis and confusing coloration skin
Nature Communications (2021)
-
Variable crab camouflage patterns defeat search image formation
Communications Biology (2021)
-
Partially coupled reaction norms of pupal colour and spot size in a butterfly
Evolutionary Ecology (2021)
-
Fear of the nighttime: substrate preference in Amazonian tadpoles
acta ethologica (2021)
Comments
Past submitting a comment you agree to abide past our Terms and Customs Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Source: https://www.nature.com/articles/s41598-019-44349-2
Posted by: palacioscaved1966.blogspot.com

0 Response to "What Type Of Defense Includes Animals That Are Colored To Match The Features Of Their Habitat"
Post a Comment